Hierarchical Thermodynamics
|
|
1998 is the 125th anniversary of the publication by J. Willard
Gibbs of his first thermodynamic work "Graphical methods in the thermodynamics
of fluids". J. Willard Gibbs later created the general thermodynamic
theory, which is a strict physical theory applying to the whole real world.
This theory has been of limited, or of questionable use in biology for
the investigation of open systems. Recently the theory has been extended
to real open biological systems and a hierarchical equilibrium thermodynamics
has been created . A study of quasiclosed systems enables one to draw conclusions
about the thermodynamic direction of biological evolution and aging of
living beings. The most essential application of the theory relates to
the study of living creature's behavior and to anti-aging medicine, gerontology,
pharmacology, nutrition and other branches of biology and medicine.
Supramolecular thermodynamics
of closed biological
systems
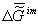 <0 <0
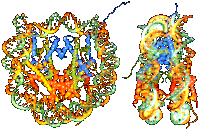 Structure
of the nucleosome Structure
of the nucleosome
Karolin Luger et al.,
Nature 389 / September 1997, p.251
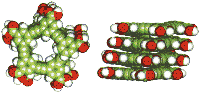
Helical conformation of a meta-substituted
phenylacetylene octadecamer (n=18), where R=H and the end groups have been
removed
J.C.Nelson et al.,
Science 277 / 19 September, 1997,
p.1793
Supramolecular thermodynamics
of quasiclosed biological systems
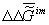 <0 <0

|
. |
|
Thermodynamics of complex hierarchical
natural systems, or macrothermodynamics, seems to focus on the three problems:
1. Whether current quasi-equilibria, that might be characterized
in terms of the corresponding thermodynamic function extremum values, may
be established in evolutionary open systems?
2. Whether quasi-closed type subsystems may be singled
out from open hierarchical systems allowing the study, in appropriate time
scales, of the behavior and evolution of the subsystems using thermodynamic
functions with extremum properties?
3. Whether behavior and evolution of open non-stationary
systems can be studied using mean specific values of classical thermodynamic
function tending to extremum?
The answers to the first two questions are trivial
and can be provided on the basis of the generally known concepts. The third
question appears to be a new one and can be answered positively when the
open system under consideration is in thermostat, together with which it
presents a complete thermodynamic system. The totality of the environment
(thermostat) and a living organism (an open non-stationary system per se)
furnishes an example of such complete system. Primarily, the non-stationary
open system under consideration is not in equilibrium with its thermostat;
its evolution is explained in terms of the tendency to extremum of the
mean specific value corresponding to the classic thermodynamic potential
of the system formation. The system evolution is directed towards partial
system - thermostat equilibrium. In the case of biological systems it is
convenient to use mean specific values of the Gibbs function related to
a unit of volume or mass, e.g. the mean specific value of the Gibbs function
for intermolecular interactions at formation of supramolecular structure
of an organism biotissue j is 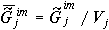 .
It has been show for the first time that in the case when there is a thermostat
which provides constancy of the environment’s chemical composition the .
It has been show for the first time that in the case when there is a thermostat
which provides constancy of the environment’s chemical composition the 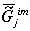 value
of an open biosystem j has the tendency to a minimum. This trend
of the value
of an open biosystem j has the tendency to a minimum. This trend
of the 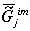 value to
a minimum explains the accumulation of a substance with chemically high
energy capacity by the biosystem which causes increase in the mean specific
chemical component value to
a minimum explains the accumulation of a substance with chemically high
energy capacity by the biosystem which causes increase in the mean specific
chemical component 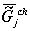 of
the biological structure during long periods of evolution. The constructiveness
of this new concept is evident because it focuses on the investigation
of the open non-stationary system characteristics per se. This is very
attractive because it offers the possibility of obtaining important quantitative
information on the basis of experimental data. This approach has enabled
us to substantiate and experimentally prove the possibility of a biosystem’s
thermodynamic characteristics being inherited during the long stages of
biological evolution when the environment remains practically unchangeable. of
the biological structure during long periods of evolution. The constructiveness
of this new concept is evident because it focuses on the investigation
of the open non-stationary system characteristics per se. This is very
attractive because it offers the possibility of obtaining important quantitative
information on the basis of experimental data. This approach has enabled
us to substantiate and experimentally prove the possibility of a biosystem’s
thermodynamic characteristics being inherited during the long stages of
biological evolution when the environment remains practically unchangeable.
|
|
REFERENCES
- G.P.Gladyshev, Thermodynamics and Macrokinetics of Natural Hierarchic
Processes (in Russian), Nauka, Moscow,1988.
- G.P.Gladyshev, Thermodynamic Theory of the Evolution of Living Beings,
NOVA Sci.Publ.,Inc.,NY, USA,1997.
- G.P.Gladyshev, "Thermodynamics
of Aging", Materials for the Symposium "Termodynamics and
Information Theory in Biology",1998.
- Differential equations of macrothermodynamics.
The systems and the processes.
|


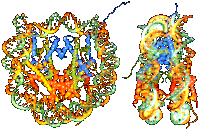 Structure
of the nucleosome
Structure
of the nucleosome 

